ASP® Titanium
This medical grade titanium ASP® needle offers maximum biocompatibility and is used in preference to treat diseases requiring long term implantation.
HOMOLOGATION
CE 0123, FDA, Health Canada, Australia
INNOVATION / OUR +
It is non-magnetic.
PACKAGING
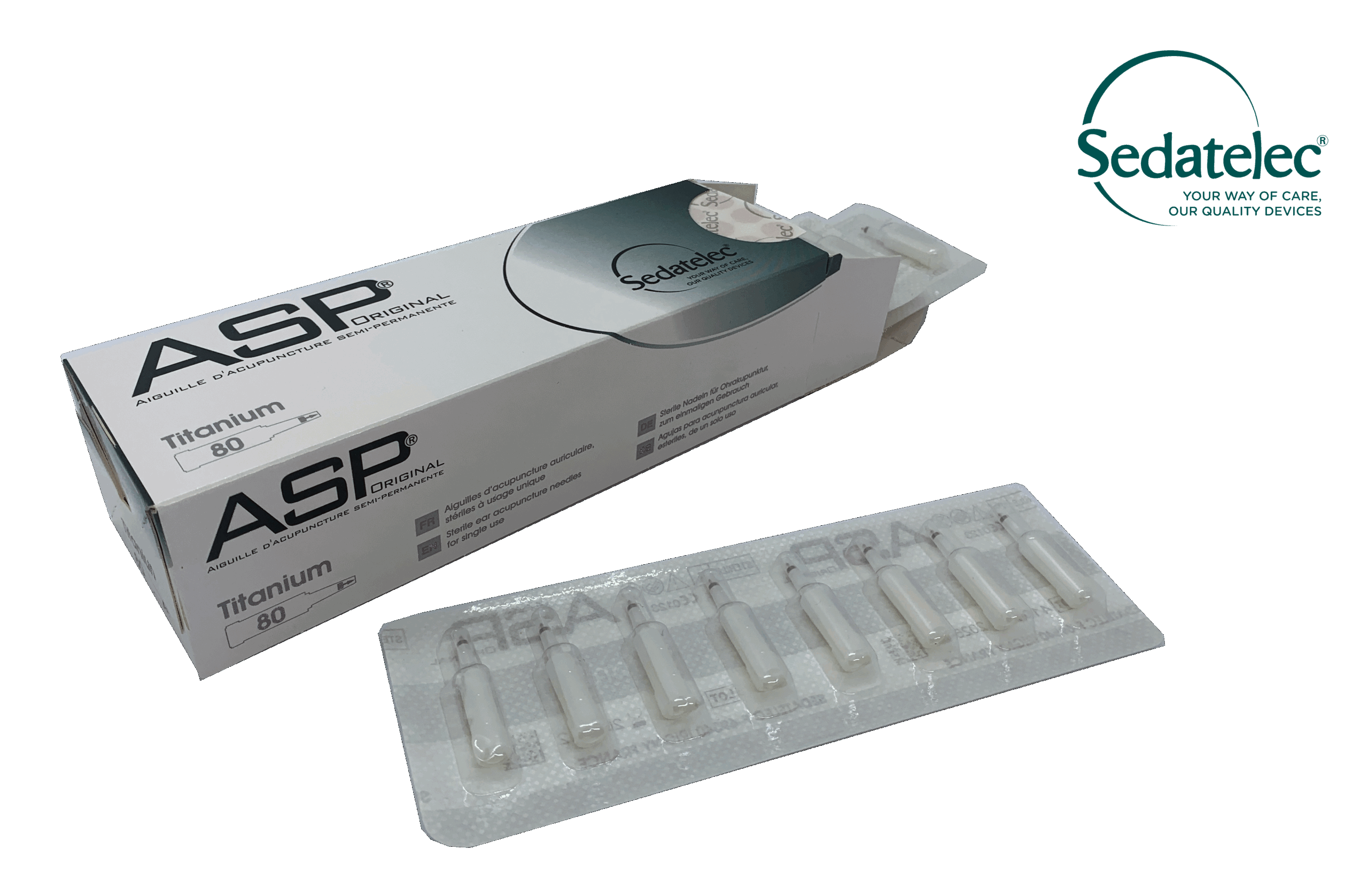
Blister packed individually and packaged in sets of 8 or 8 needles.
STERILISATION
Sterilised by Gamma radiation (5 year shelf life).
Medical device IIa class.
PRESENTATION
This medical grade titanium ASP® needle offers maximum biocompatibility and is used in preference to treat diseases requiring long term implantation. It is highly recommended for at risk patients: diabetic patients and those with immunodeficiencies or multiple allergies.
The indisputable advantages of the ASP® Titanium Original:
- Rapid, accurate application with no risk of infection.
- Original shape allowing prolonged stimulation of the auricular point (for up to a few days).
- Hypoallergenic self-adhesive plasters supplied in each box to protect the needle.
- The Sedatelec «click» demonstrates the irreproachable quality of the injector, the result of more than 40 years of experience.
- Manufacture by Sedatelec in France in clean rooms, perfect quality and excellent behaviour of materials over time.
Product references :
110638 ASPT8 – ASP Original TITANIUM 8
110626 ASPT80 – ASP Original TITANIUM 80
The adhesive plasters supplied with the ASP® are manufactured with a micro-porous support and a hypoallergenic adhesive film.
They are used as required to hold the needle in place and to protect the head of the needle.
Available in bags of 600 plasters, reference : 103621 – ASPLAST600.
Before use, carefully read the user manual.
OrderPRODUCT VIDEO
Testimony
John Howard